Phytophthora cinnamomi
The fungus forms the following morphological structures:
• asexual: sporangia, chlamydospores
• sexual: oospores, antheridia, oogonia
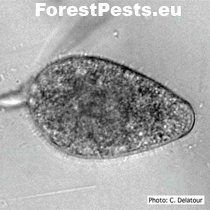
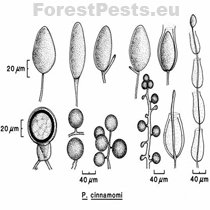
The sporangia are ovoid, pear-shaped, elliptical to elongated elliptical with a discreet thickening of the sporangium tip; it belongs to the group of non-papillate Phytophthora. They are narrowed or round at the base, non-deciduous from the mycelium. They are formed at the end of the mycelium. New sporangia are formed by internal or external sags or sympodial branching of sporangiophore just below the empty sporangium (mycelium is divided into two branches by peak growth, later divided into two branches, etc.).
The spore size is 75x40 µm with a length-to-width ratio of 1.54. Sporangia are poorly formed in oxygen-free cultures. However, they form very well when the mycelial discs on the nutrient medium are placed in an unsterilized soil solution (10 g of soil per 1 l of water) or washed with saltwater.
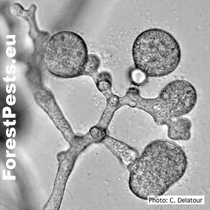
Chlamydospores are formed in large numbers in cultures and infected tissues on the maternal mycelium and new mycelial branches. They are spherical with a thin wall with a diameter of 31 to 50 µm. They often occur in clusters of up to 10 pieces in the shape of a grape bunch. Oospores, oogonia, and antheridia form the sexual structures. The fungus is heterothallic, i.e., two generically different mating types "pairing types A1 and A2". Only when the mycelia of opposite pairing types merge the formation of oospores happen. It occurs mainly in mating type A2 in nature, so it is primarily a homothallic species.However, oospores are sometimes formed in cultures of the A2 type, e.g., on barley grains or in the presence of the fungus Trichoderma viride.
The oogonia are round, often with a level base, with a smooth surface, translucent to yellowish-brown with a diameter of 21 - 58 µm (average 40 µm).
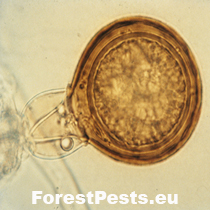
The oospores are round, translucent, even yellow-brown, plerotic (i.e., there is no free space between the oospore and the oogonia, the oospore fills the entire oogonia). The size of the oospore is 19 - 54 µm (with a diameter of 20 µm).
The fungus grows at a temperature of 5 °C to 34 °C with an optimum of 24 - 28 °C.
 |
 |
 |
|
Eucalyptus marginata dieback in Austrálii.
Foto: E.Hansen http://forestphytophthoras.org/
species/cinnamomi)
|
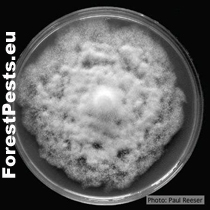
Pure culture of Phytophthora cinnamomi. Photo: P. Reeser.
http://forestphytophthoras.org/species/cinnamomi
|
Ovoid non-papillate sporangia in water. Foto: C.Delatour.
http://forestphytophthoras.org/species/cinnamomi
|
Bionomy - infection cycle:
The fungus causes infection of various woody plants' roots and root necks, even some monocotyledonous grasses in the size of trees (Xanthorrhoea australis). After the colonization of the root, symptoms of infection appear on the assimilation organs (leaves, needles), first their chlorosis, wilting to necrosis. The higher the air temperature in the ecosystem, the faster the development of the disease and symptoms. In the cold conditions of the mountain valleys, the symptoms develop very slowly, and the infested trees survive for several years. Infected trees, but still without signs of infection on assimilation organs, are the most dangerous sources of spreading the pathogens to new localities, especially from the forest and ornamental tree nurseries.
The fungus P. cinnamomi has an extraordinary ability to survive in nature in the saprophytic state than other species of the genus Phytophthora. It survives in moist soil for up to 6 years, not as a mycelium but in the form of chlamydospores. Mycelium is relatively quickly decomposed by soil microorganisms, especially antagonistic fungi of Trichoderma spp. and Gliocladium spp.
The disease develops at 15 - 25 °C and rapidly slows at 30 °C and higher. The intensity of the disease symptoms increases significantly during heavy rainfall or excessive irrigation. The disease rarely develops at temperatures below 12 ° C. When the environmental conditions are suitable for tree growth (i.e., in ecological optimum of trees), the fungus may be present but does not cause disease to the tree; respectively, the infection is not so severe that also the symptoms on the tree are visible.
• tree stress caused by drought or wetting
• shallow soil, under which there is an impermeable layer of clayey soil or rocks
• poor drainage of excess water around the trees
• lack of organic matter in the soil; leads to insufficient production of microbial life in the soil.
Endangered woody plants:
There is a wide range of deciduous and coniferous forest trees and fruit trees endangered in the world. The pathogen infects almost all woody plants in suitable climatic conditions (warm climatic conditions).
Very susceptible woody plants are Ananas comosus and Persea americana.
Medium-susceptible woody plants are Castanea sativa, Cinnamomum verum, Cupressaceae, Ericaceae, Eucalyptus spp., Fagus sylvatica, Juglans regia, Kalmia latifolia, Pinaceae (e.g., Pinus radiata), Platanus spp., Quercus spp., Rhododendron hybrids.
It occurs on Castanea sativa in Slovakia; the first finding was in 1972.
 |
 |
 |
|
Mycelial lumps and chlamydospores in grape-like clusters Photo: C. Delatour. http://forestphytophthoras.org
|
Bottom left connection of antheridia to oogonium, amphigynous, obvious plerotic oospore. |
Chlamydospores and mycelial lumps, the proliferation of sporangium (overgrowth of mycelia through empty sporangium) |
Damaged plant parts: Root (thin roots and main supporting roots), root neck, and trunk base.
Significance: Very significant
Origin and distribution:
The fungus comes from tropical and subtropical regions, mainly from Australia. Climate warming creates suitable conditions for the life of this fungus also in Europe, especially in the southern areas, where the winter is mild.
Endangered stands and areas:
Beech stands are particularly endangered (31.6% forests of cover, 632 thousand ha). The pathogen can be transmitted to the forest from gardens where ornamental woody plants died (e.g., rhododendrons, etc.) and were carried as a waste to the forest "where they later rot". Thus, the first diebacks can be observed next to houses and residential areas in the woods. Gardens are intensively maintained - dead plants are replaced with new ones, mainly bought in gardening supplies, where plants are imported from abroad and from regions where the disease is common.
Prevention:
Plant the seedlings in the disease-free stand, not only without symptoms but also without, e.g., the most resistant structures of the fungus, chlamydospores.
It is necessary to prepare a functional system (protocol) of plant growing hygiene in the production of seedlings in nurseries and ensure strict compliance.
The stand is about integrated protection, i.e., a combination of cultivation, biological and chemical methods.
Among the chemical substances, metalaxyl, salts of phosphoric acid (e.g., fosetyl-Al, and the others), or phenylamide show biological (fungicidal and fungistatic) efficiency.
Among biologically active organisms, fungi's antagonistic properties from the genera Trichoderma and Gliocladium are effective.
The water used for watering and irrigating must be disinfected, e.g., chlorine.
Growing methods include a positive selection of tree species or clones resistant to this pathogen, e.g., Juniperus virginiana. Woody plants that tolerate infection (trees are infested but survive without significant signs of damage) include Pinus spp., Picea spp., Larix spp.
Highly susceptible woody plants to infection are Tsuga heterophylla, Tsuga mertensiana, Pseudotsuga menziessii, and Abies spp.
Monitoring:
Usually, from the stump up to about 2 m in the height of the trunk, tar-like stains are searched. These spots usually occur on the tree, showing chlorosis, small leaves, leaves necrosis, and defoliation. Below the bark periderm, there are apparent changes in the color of the tissues. Next, a sample of about 2 cm is taken to the laboratory to isolate the pathogen. The final determination depends on the microscopic properties of sporangia, zoospores, oogonia, antheridia, and oospores. Genetic analyzes are also used, but usually, the pathogen must be isolated in pure culture, and to start the analysis, it is necessary to have multiple samples collected (due to the economics of the analysis). Trees of all ages can be attacked, from seedlings to centuries-old trees.
 |
|
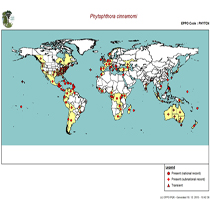
Distribution of Phytophthora cinnamomi by EPPO records as of 1.10.2015. Photo: www.eppo.int . |
Pest control:
Forest protection against this fungus is problematic because:
• it has a wide range of host trees,
• can infect the plant and survive for a long time on it without showing any signs of infection on the plant,
• survives for six years in soil, roots, and dead plant material in the form of chlamydospores, and fumigation does not always reach such a depth in the soil that these chlamydospores would be inactivated.
If possible, fumigation with methyl bromide is effective, e.g., in forest and ornamental tree nurseries, where it is possible to apply the product even deeper into the soil. Disinfect irrigation water by oxidation, e.g., chlorine preparations.
Pest category: Fungi
Found in Slovakia: Yes
Invasive species: Yes
Similar species: All species of the genus Phytophthora, and also Pythium.
Bharath, S., Phillips W., Krauss, U., 1999: Practical Notes on Work with Phytophthora Species. In: Krauss, U. and Hebbar, P., Research Methodology in Biocontrol of Plant Diseases with Special Reference to Fungal Diseases of Cocoa, Workshop Manual, CATIE, Turrialba, Costa Rica, p.123-130.
Brasier, C. M., Rose, J., Gibbs, J. N., 1995: An unusual Phytophthora associated with widespread alder mortality in Britain. Plant Pathology, 44: 999-1007.
Brasier, C., 2007: Phytophthora Biodiversity: How Many Phytophthora Species Are There? In: Proceedings from the Forth Meeting of IUFRO Working Party S07.02.09, Phytophthoras in Forests and Natural Ecosystems, August 26 – 31, 2007, Monterey, California, p. 101 – 115.
Day, W. R., 1938: Root-rot of sweet chestnut and beech caused by species of Phytophthora. I. Cause and symptoms of disease: its relation to soil conditions. Forestry, Oxford, 12: 101-116.
Day, W. R., 1939: Root-rot of sweet chestnut and beech caused by species of Phytophthora. II. Inoculation experiments and methods of control. Forestry, Oxford, 13: 46-58.
Doster, M.A., Bostock, R.M., 1988: Susceptibility of almond cultivars and stone fruit species to pruning wound cankers caused by Phytophthora syringae. Plant Disease, 72: 490-492.
El-Hamalawi, Z.A., Menge, J.A., 1994: Effects of wound age and fungicide treatment of wounds on susceptibility of avocado stems to infection by Phytophthora citricola. Plant Disease, 78: 700-704.
Erwin, D.C., Ribeiro, O.K., 1996: Phytophthora Disease Worldwide. APS Press, St. Paul, Minnesota, 562 pp.
Gibbs, J. N., 1995: Phytophthora Root Disease of Alder in Britain. EPPO Bulletin, 6 pp.
Gibbs, J., 1994: Phytophthora Root Disease of Common Alder. Forestry Commission, Research Information Note, 258, 4 pp.
Gibbs, J., Lonsdale, D., 1996: Phytophthora Disease of Alder: the situation in 1995. Forestry Commission, Research Information Note, 277, 4 pp.
Hartmann, G., Blank, R., 1998: Buchensterben auf zeiweise nassen Standorten unter Beteiligung von Phytophthora-Wurzalfäule. Forst und Holz, 53 (7): 187-193.
Hartmann, G., Blank, R., 2002: Vorkommen und Standortbezüge von Phytophthora-Arten in geschädigten Eichenbeständen in Nordwestdeutschland (Niedersachsen, Nordrhein-Westfalen und Schleswig-Holstein). Forst und Holz, 57 (18): 539-545.
Juhásová, G., 1999: Hubové choroby gaštana jedlého (Castanea sativa Mill.). Veda, Bratislava, 191 pp.
Juhásová, G., Satko, J., 1997: Zdravotný stav Castanea sativa Mill., výskyt huby Cryphonectria parasitica (Murr.)Barr a možnosti biologickej ochrany na Slovensku. In: Pestovanie a ochrana gaštana jedlého (Castanea sativa Mill.) na Slovensku, Zborník, Ústav ekológie lesa SAV Zvolen, Pobočka biológie drevín Nitra, Nitra, 17 - 35.
Jung, T., Blaschke, H., 1995: Phytophthora root rot in declining forest trees. Phyton (Horn, Austria), 36 (3): 95-102.
Jung, T., Blaschke, H., Neumann, P., 1996: Isolation, identification and pathogenecity of Phytophthora species from declining oak stands. Eur. J. For. Path., 26: 253-272.
Jung, T., Cooke, D. E. L., Blaschke, H., Dunkan, J. M., Oßwald, W., 1999: Phytophthora quercina sp. nov., causing root rot of European oaks. Mycol. Res., 103 (7): 785-798.
Jung, T., Hansen, E. M., Winton, L., Oßwald, W., Delatour, C., 2002: Three new species of Phytophthora from European oak forests. Mycol. Res., 106 (4): 397-411.
Kunca, A., 2003: Phytophthora ako vážny patogén buka lesného v Nemecku. In. Varínsky, J. (Ed.), Zborník referátov z celoslovenského seminára Aktuálne problémy v ochrane lesa 2003, Banská Štiavnica, 24-25.4.2003, p. 175-178.
Lilja, A., Rikala, R., Hietala, A., Heinonen, R., 1996: Stem lesions on Betula pendula seedlings in Finnish forest nurseries and the pathogenecity of Phytophthora cactorum. Eur. J. For. Path., 26: 89-96.
Mitchell, D.J., Kannwischer-Mitchell, M.E., 1992: Phytophthora. In: Singleton, L.L., Mihail, J.D., Rush, C.M. (eds), Methods for Research on Soilborn Phytopathogenic Fungi, APS Press, St. Paul, Minnesota, p. 31-38.
Robin, C., Smith, I., Hansen, E.M. 2012. Phythophthora cinnamomi. Forest Phytophthoras 2(1). doi: 10.5399/osu/fp.2.1.3041
Sánchez, M. E., Caetano, P., Ferraz, J., Trapero, A., 2002: Phytophthora Disease of Quercus ilex in south-western Spain. Forest Pathology, 32 (1): 5-18.


