Mycosphaerella pini
Dothistroma (Red band needle) blight is one of the most severe diseases of the assimilative organs of pines. This pathogen was introduced to Slovakia in the mid-90s of the 20th century.
Mycosphaerella pini is the teleomorphic stage of the fungus Dothistroma septosporum (Dorog) Morelet. The sexual stage of fungus Mycosphaerella pini Rost. in Munk is rarely occurring in our conditions.
The first symptoms are pale yellow to brown spots, reminiscent of insect wounds. In November, the infected needles are already brown. By the end of December, brownish-red spots appear on the browned needles up to the transverse stripes (hence the name "Red band needle"). Pycnidia are black pustules that pierce the epidermis, forming conidia in the spring.
 |
|||
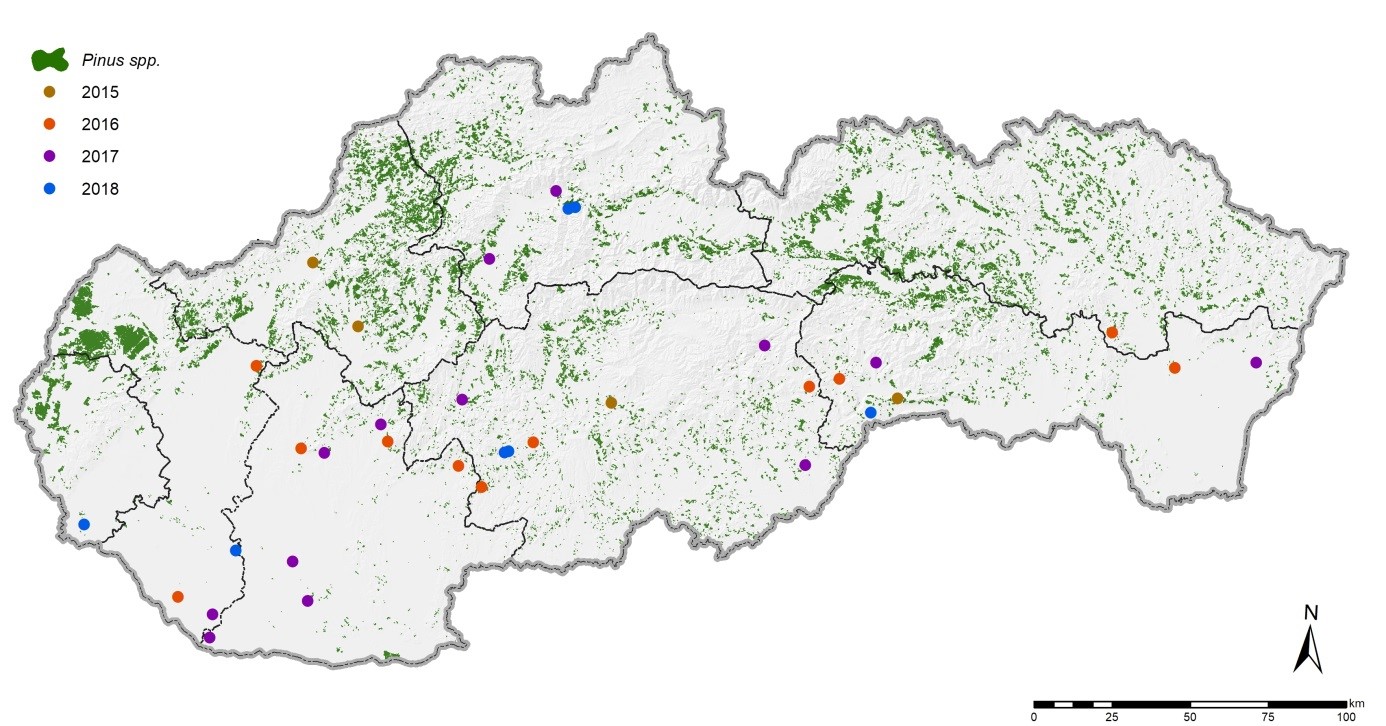
| Map of Pinus nigra sites where the presence of Mycosphaerella pini on assimilation organs was confirmed (2015 - 2018) |
Prevention:
Found in Slovakia: Yes
Invasive species: Yes
Similar species:
Some species of genus Lophodermium cause similar symptoms on pines in our conditions. One of the identifying features is a red band on the needles when attacked by Dothistroma (Red band needle) blight.
Adamčíková, K., Hečková, Z., Ondrušková, R., 2015: Ochorenie borovíc spôsobené hubami Dothistroma septopora a Dothistroma pini. In: Aktuálne problémy v ochrane lesa 2015, zborník referátov z medzinárodnej konferencie, ktorá sa konala 29. a 30. januára 2015 v Novom Smokovci, NLC Zvolen, s. 83–87.
Barnes, I., Kirisits, T., Wingfield, M. J., & Wingfield, B. D., 2011: Needle blight of pine caused by two species of Dothistroma in Hungary. Forest Pathology 41: s. 361–369.
Barnes, I., Cortinas, M. N., Wingfield, M. J., Wingfield, B. D., 2008: Microsatellite markers for the red band needle blight pathogen, Dothistroma septosporum. Molecular Ecology Resources 8: s. 1026–1029.
Barnes, I., Crous, P. W., Wingfield, M. J., Wingfield, B. D., 2004: Multigene phylogenies reveal that red band needle blight of Pinus is caused by two distinct species of Dothistroma, D. septosporum and D. pini. Studies in Mycology. 50: s. 551–565.
Bassett, C., 1969: Larix decidua a new host for Dothistroma pini. Plant Disease Reporter 53: s. 706.
Bednářová, M., Palovčíková, D., Jankovský, L., 2006: The host spectrum of Dothistroma needle blight Mycosphaerella pini E. Rostrup – new hosts of Dothistroma needle blight observed in the Czech Republic. Journal of Forest Science 52: s. 30–36.
Bednářová, M., Bodejčková, I., Palovčíková, D., Jankovský, L., 2007: The Contemporary Situation of Dothistroma Needle Blight Outbreak in the Czech Republic. Acta Silvativa and Lingaria Hungarica, Spec. Edition s. 17–21.
Bradshaw, R.E., 2004: Dothistroma (red-band) needle blight of pines and the dothistromin toxin: a review. Forest Pathology 34: s. 163–185.
Brown, A.,– Rose, D., Webber, J., 2003: Red band needle blight of Pine. Forestry Commission Edinburgh: Information note.
Butin, H., Richter, J., 1983: Dothistroma – Nadelbräune, eine neue Kiefernkrankheit in der Bundesrepublik Deutschland. Nachr.- -Bl. Dtsch. Pfl.-Schutzdients 35: s. 129–131.
Drenkhan, R., Adamson, K., Jürimaa, K., Hanso, M., 2014: Dothistroma septosporum on firs (Abies spp.) in the northern Baltics. For. Path. 44: s. 250–254.
Dubin, H.J., Walper, S., 1967: Dothistroma pini on Pseudotsuga menziesii. Plant Disease Reporter 51: s. 454.
Gibson, I.A.S., 1972: Dothistroma blight of Pinus radiata. Annual Review of Phytopathology (10): s. 51–72.
Ioos, R., Fabre, B., Saurat, C., Fourrier, C., Frey, P., Marcais, B., 2010: Development, comparison, and validation of real-time andconventional PCR tools for the detection of the fungal pathogens causing brown spot and red band needle blights of pine. Phytopathology 100: s. 105–114.
Jankovský, L., Bednářová, M., Palovčíková, D., 2004: Dothistroma needle blight Mycosphaerella pini E. Rostrup, a new quarantine pathogen of pines in the CR . Journal of Forest Science 50: s. 319–326.
Jankovský, L., 2007: Červená sypavka borovice Mycosphaerella pini E. Rostrup běžným patogenem? Lesnická práce 16: s. 12.
Karadzić, D., 1989: Scirrhia pini. Life cycle of the fungus in plantations of Pinus nigra in Serbia. European Journal of Forest Pathology 19: s. 231–236.
Karadzić, D., 1988: Dothistroma septospora (Dorog.) Morelet: rasprostranjenje, biljke domacini i znacaj [Dothistroma septospora: distribution, hosts and importance]. Glasnik Sumarskog Fakulteta, Univerzitet u Beogradu 70: s. 133–144.
Kapitola, P., Růžička, T., Kroutil, P., 2011: Karanténní škodlivé organismy na lesních dřevinách. Státní rostlinolékažská správa Praha. 63 pp.
Kowalski, T., Jankowiak, R., 1998: First record of Dothistroma septospora (Dorog.) Morelet in Poland: a contribution to the symptomology and epidemiology. Phytopatologia Polonica 16: s. 16–29.
Krstić, M., 1958: Nezabeležene fitopatološke pojave u rasadnicima i šumama Srbije. [Unnoted phytopathological phenomena in nurseries and forests of Serbia]. Zaštita bilja 45: s. 75–79.
Kunca, A., Foffová, E., 2000: Ohrozenie porastov borovice čiernej fytokaranténnym patogénom Dothistroma septopora (Dorog). Morelet. In: Aktuálne problémy v ochrane lesa 2000, zborník z referátov z celoslovenského seminára, 17.–18. apríla 2000 v Banskej Štiavnici, Zvolen, LVÚ Zvolen, s. 136–139.
Landmann, G., 2000: Forest health in France: assessment for 1998 and new facts. Revue Forestiere Francaise 52: 9–22.
Murray, J.S., Batko, S., 1962: Dothistroma pini Hulbary: A new disease on pine in Britain. Forestry 34: s. 57–65.
Parker, A.K., Collis, D.G., 1966: Dothistroma needle blight of pines in British Columbia. The Forestry Chronicle 42: 160-161.
Petrak, F., 1961: Die Lecanosticta Krankheit der Föhren in Österreich. Sydowia 15: 252–256.
Szabó, I., 1997: A Dothistroma septosporum (Dorog.) Morlet fellése feketefenyö ültetvényeken [Occurrence of Dothistroma septosporum in Austrian pine plantations]. Erdészeti Lapok 132: s. 44–45.
Watt, M. S., Kriticos, D. J., Alcaraz, S., Brown, A. V., Leriche, A., 2009: The hosts and potential geographic range of Dothistroma needle blight. Forest Ecology and Management 257: s. 1505–1519.
Woods, A., 2011: Is the health of British Columbia’s forests being influenced by climate change? If so, was this predictable? Canadian Journal of Plant Pathology 33: s. 117–126.
Zúbrik, M., Kunca, A., Turčani, M., Vakula, J., Leontovyč, R., 2006: Invasive and quarantine pests in forests in Slovakia. OEPP/EPPO Bulletin 36: s. 402–408.






